Titanium Information
|
|
There are 26 known
isotopes of titanium ranging from Ti-38 to Ti-63. 46Ti (8.25%), 47Ti (7.44%),
48Ti, (73.72%), 49Ti (5.41%) and 50Ti (5.18%) are the five stable isotopes. The
most naturally abundant of these isotopes is 48Ti at 73.7%. Titanium is the ninth most abundant metal in
Earth’s crust!
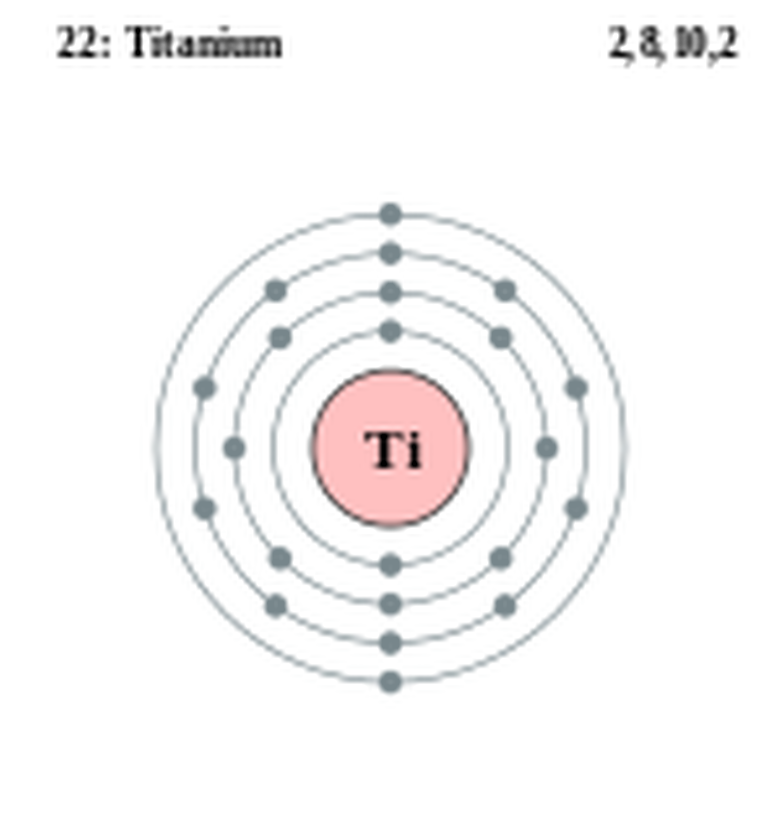
Titanium has 22 electrons, 22 protons, and 26 neutrons in its most abundant isotope. The element has two valence electrons. A valence electron is It can make +4, +3, or +2 ions. It has four shells and there are 2 electrons in the first energy shell, 8 in the second, 10 in the third shell and 2 in the last shell. |